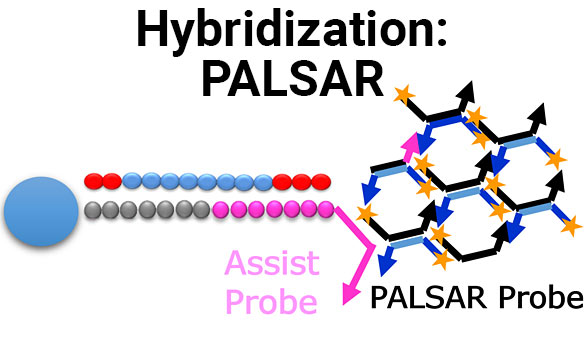
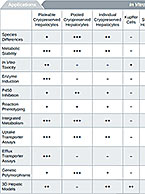
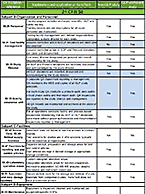
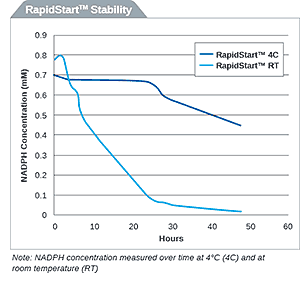
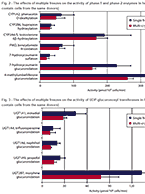
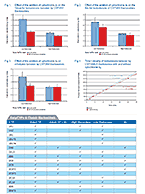
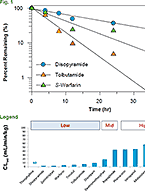
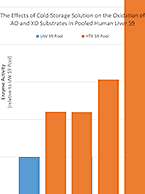
Drug metabolism studies profile metabolic pathways and likely metabolites formed by a compound, as well as identify drug-metabolizing enzymes involved in its biotransformation. For more information about these services, visit our Drug Metabolism contract studies page.